
Low-energy extracorporeal shock wave therapy as a treatment for greater trochanteric pain syndrome.
Furia JP, Rompe JD, Maffulli N.
Am J Sports Med 2009;37:1806-1813
This case-control study by Furia JP, Rompe JD, Maffulli N. aimed to determine whether low-energy RSWT® is a safe and effective management modality for chronic greater trochanteric pain syndrome.

Methods
GTPS was defined as symptoms of moderate-to-severe pain located over the greater trochanter and peritrochanteric area, pain with resisted hip abduction, and impaired function.
The study cohort consisted of two carefully selected groups:
- Shock Wave Therapy (SWT) group (N=17) - patients with an established diagnosis of chronic GTPS treated with Radial Shock Wave Therapy (RSWT®);
- Control group (N=15) - a similar cohort of patients with GTPS who were treated in the same time interval with additional forms of traditional nonoperative methods but did not receive RSWT®.
There was no statistical difference (P >0.05) in age and duration of symptoms between the studied groups.
Each patient from the SWT group received one low-energy treatment session of RSWT® without anesthesia. Investigator applied 2,000 impulses (air pressure of the device set at 4.0 bar, EFD = 0.18 mJ/mm2). The treatment frequency was 10 shocks/s. The total energy flux density of the treatment session was approximately 360 mJ/mm2.
Radial extracorporeal shock waves were administered using the DolorClast® Radial Shock Waves.
Outcomes
All participants received a detailed pretreatment evaluation and were asked to participate in a follow-up examination (1, 3, and 12 months post-treatment), which included:
- Visual Analog Scale (VAS) to assess pain,
- Harris Hip Score (HHS) to grade pain, function, range of motion, and deformity associated with hip dysfunction,
- The modified Roles & Maudsley (RM) score to evaluate the patient's pain affecting normal daily activities.
Results
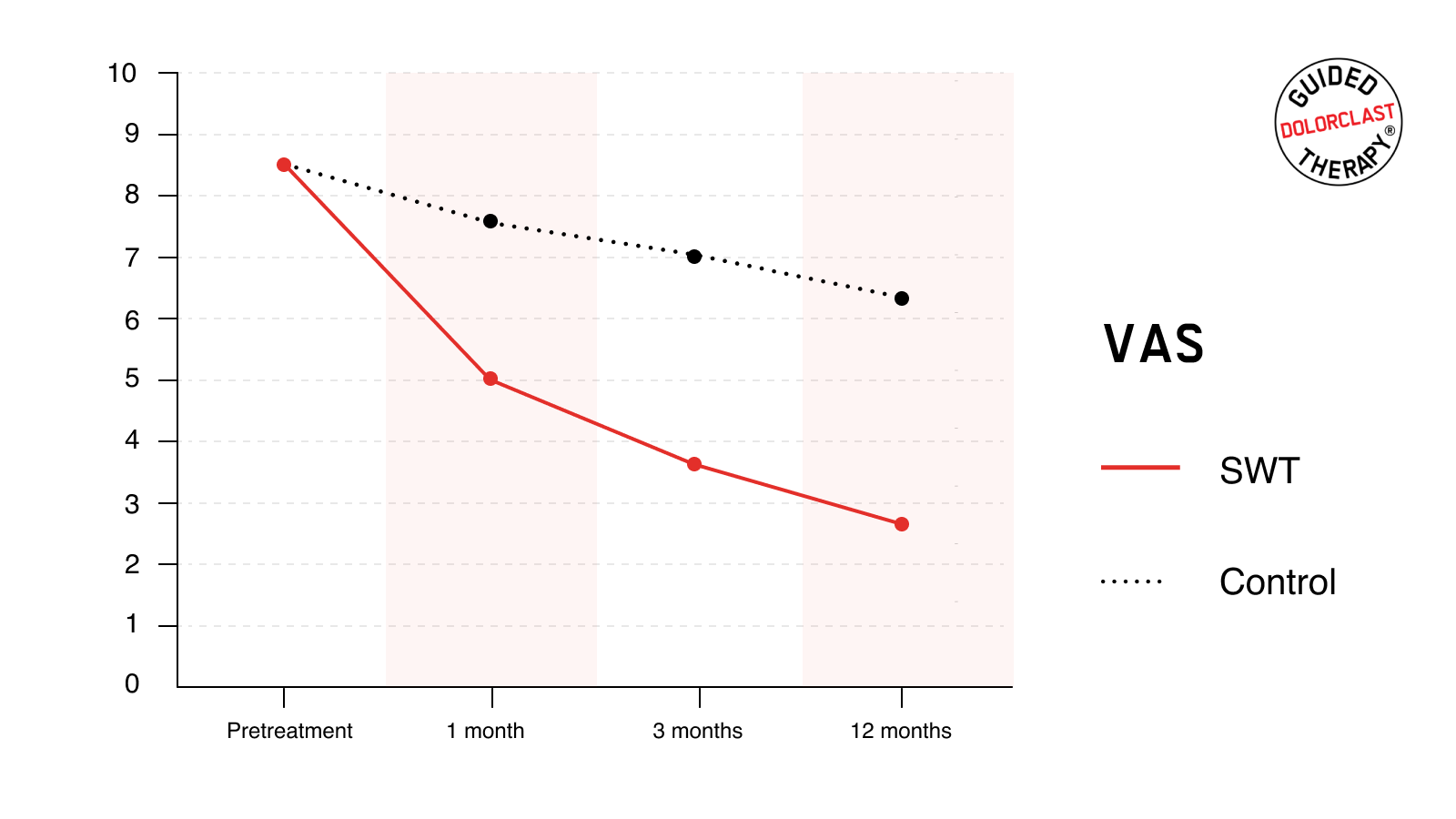
VAS
The mean pretreatment VAS score for both SWT and the control group was 8.5 ± 0.9. RSWT® led to statistically significant pain relief. One month after treatment, the mean VAS score decreased to 5.1 ± 0.9. Pain reduction was even more pronounced 3 and 12 months following therapy; the mean VAS score decreased to 3.7 ± 0.8 and 2.7 ± 0.9, respectively. In contrast, such results were not observed in the control group. The baseline result was reduced to 7.6 ± 1.0, 7.0 ± 0.8, and 6.3 ± 1.2. For each follow-up time point, the degree of the improvements in VAS was significantly greater for the SWT than for the control group (P <0.001).
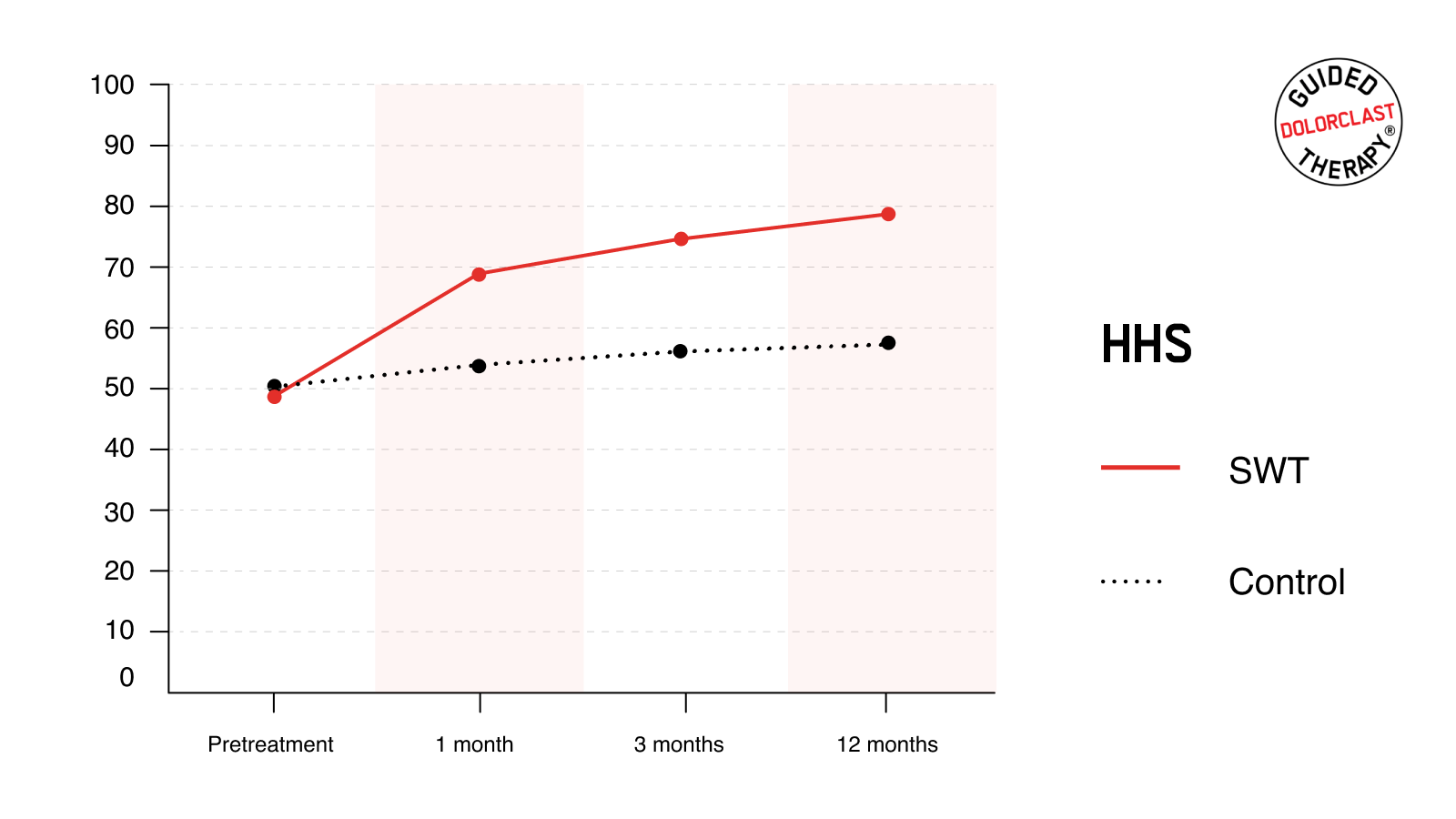
HHS
The mean pretreatment HHS for the SWT and control group was 49.6 ± 4.9 and 50.4 ± 4.4, respectively. Individuals treated with RSWT® reported a substantial and lasting improvement; mean HHS increased to 69.8 ± 7.3, 74.8 ± 5.9, and 79.9 ± 6.2, as assessed 1, 3, and 12 months after the treatment, respectively. Likewise, these improvements were not comparable within the control group. After the treatment, HHS increased slightly but oscillated at a similar level (54.4 ± 5.0, 56.9 ± 5.2, and 57.6 ± 5.8). For each follow-up point, the magnitude of the improvements in HHS was statistically greater for the SWT than for the control group (P <0.001).
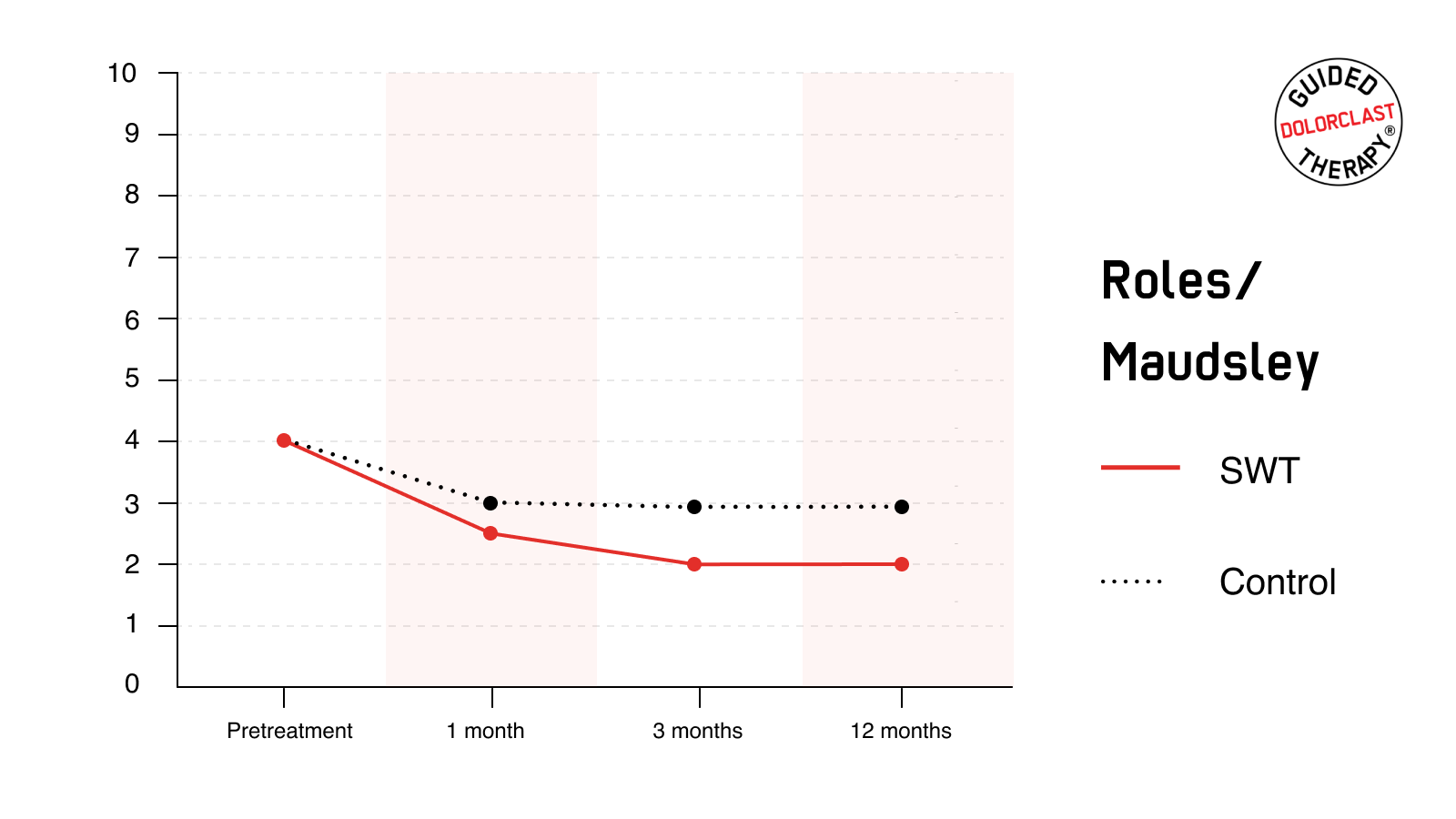
RM
At the baseline evaluation, SWT and the control group rated the condition of the affected hip as 4 (indicating poor status). RM score decreased in both groups. However, results were considerably more pronounced in individuals receiving RSWT®. The proportion of patients with excellent or good RM scores (i.e., successful results) in the follow-up assessment was statistically (P<0.001) greater at each time point in the SWT group than in the control group. Most importantly, satisfactory improvement was maintained for at least one year, as reflected by good or excellent condition reported by 79% of individuals treated with RSWT®.
Conclusion
Given the lengthy nature of the traditional treatment, questionable results, and common recurrence, DolorClast® Radial Shock Wave Therapy is a valuable and highly effective tool in the management of GTPS. This paper provides yet another substantial scientific proof underpinning the safety and efficacy of RSWT® in managing complex musculoskeletal disorders, especially in patients with demanding physical activity.
Read the full study: http://www.ncbi.nlm.nih.gov/pubmed/19439756
