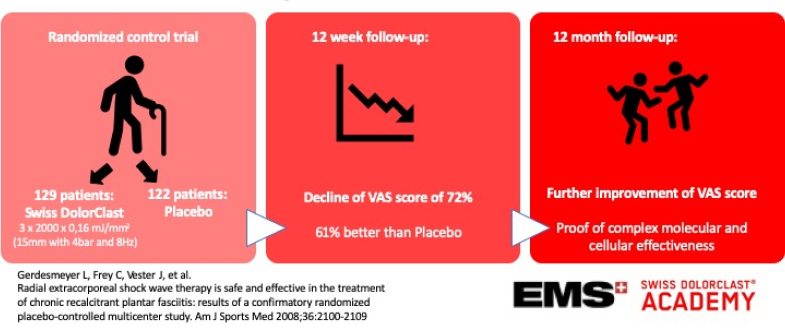
Radial extracorporeal shock wave therapy is safe and effective in the treatment of chronic recalcitrant plantar fasciitis: results of a confirmatory randomized placebo-controlled multicenter study.
Background
Radial extracorporeal shock wave therapy is an effective treatment for chronic plantar fasciitis that can be administered to outpatients without anesthesia. This randomized controlled trial aims at demonstrating therapeutic superiority vs. placebo.
Hypothesis
There is no difference in effectiveness between radial extracorporeal shock wave therapy and placebo in the treatment of chronic plantar fasciitis.
Methods
Three interventions of radial extracorporeal shock wave therapy (0.16 mJ/mm2; 2000 impulses) compared with placebo were studied in 245 patients with chronic plantar fasciitis. Primary endpoints were changes in visual analog scale composite score from baseline to 12 weeks’ follow-up, overall success rates, and success rates of the single visual analog scale scores (heel pain at first steps in the morning, during daily activities, during standardized pressure force). Secondary endpoints were single changes in visual analog scale scores, success rates, Roles and Maudsley score, SF-36, and patients’ and investigators’ global judgment of effectiveness 12 weeks and 12 months after extracorporeal shock wave therapy.
Results
Radial extracorporeal shock wave therapy proved significantly superior to placebo with a reduction of the visual analog scale composite score of 72.1% compared with 44.7% (P = .0220), and an overall success rate of 61.0% compared with 42.2% in the placebo group (P = .0020) at 12 weeks. Superiority was even more pronounced at 12 months, and all secondary outcome measures supported radial extracorporeal shock wave therapy to be significantly superior to placebo (P < .025, 1-sided). No relevant side effects were observed.
Conclusion
Radial extracorporeal shock wave therapy significantly improves pain, function, and quality of life compared with placebo in patients with recalcitrant plantar fasciitis.
Full study herehttp://www.ncbi.nlm.nih.gov/pubmed/18832341

